HOME
Interpretation
of the Chest X- Ray
A System for
looking at the film
1.
Name and side marker
2. Film
quality
Is an indication of how precise
is the
information
on the film
Penetration
A good film will allow
you to
localise one or two thoracic disc spaces.
Inspiration.
Following good
inspiration the
diaphragm is normally at or near the level of the posterior 10th
rib. The right diaphragm is usually higher than the left.
AP or PA, supine or
erect The
radiographer
should mark how the film has been taken. AP films have the scapula and
magnified heart projected over the lung fields and heart size cannot be
assessed. Most portables and all supine films are AP. Supine films show
less of
the lung fields and the mediastinum appears wide: there is no point in
trying
to decide if a patient has an aortic dissection on a supine film.
Interpretation of a film of a patient with a marked kyphosis suffers
from
similar difficulties.
Centring Allow
for rotation before evaluating
mediastinal and tracheal shift by examining the relationship between
the medial
ends of the clavicle and the posterior spinous process.
The
side to which the patient is rotated usually becomes
more translucent (black). But there are exceptions and quite marked
degrees of
rotation are required to produce significant changes in radiographic
density.
Fig 1
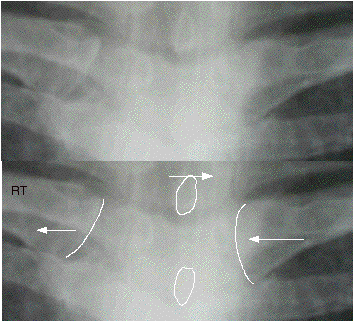 This
shows a patient who is rotated (turned towards) her right. The medial
ends of
the clavicles and the spinous processes have been outlined in the lower
picture
with their apparent direction of movement. The
mediastinum and
trachea being anterior structures will move in the same direction as
the
clavicles.
This
shows a patient who is rotated (turned towards) her right. The medial
ends of
the clavicles and the spinous processes have been outlined in the lower
picture
with their apparent direction of movement. The
mediastinum and
trachea being anterior structures will move in the same direction as
the
clavicles.
3.
Bones
Start
from outside i.e. humeri, shoulders and clavicles,
any secondaries, fractures or arthritis. Is the spine straight?
Ribs
a.
posterior, remembering to look through the heart to
where the ribs meet the spine.
b.
lateral chest wall.
c.
anterior end of each rib.
All
the time compare the two sides. Many prefer to examine
each rib as a whole but it is possible to miss one unless care is
taken.
4.
Soft Tissues
Briefly
examine the muscles of the chest wall -any surgical
emphysema?
Trachea
central? (Can be slightly to the right in patients
with a degree of aortic dilatation).
Mediastinum
and hila - size and shape and position
Diaphragm
and below - any significant discrepancy of
height. Exclude pneumoperitoneum
5.
lung fields
Compare
the two sides by mentally dividing them into 6
parts i.e. rt. and left. upper mid and lower zones. Go back to the
difficult
areas: apices, costophrenic angles, hila and through the heart. There
is a lot
of lung behind the heart.
Note
the position and thickness of the horizontal fissure
which normally extends out from the centre of the right hilum.
6. Mediastnum
Cardiac shadow
On a PA film the
maximum
width of the heart shadow compared with the maximum width of the
combined lung
fields is a good approximation to the CT ratio, normally below 50% it
can be up
to 60% in elderly patients. The true CT ratio is the sum of maximum
distances
from the centre of the heart shadow to each heart edge over the maximum
external bony thoracic wall.
Cardiac shadow
fig 2
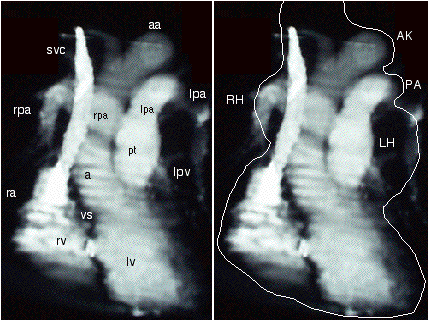
A 3D CT cardiac angiogram. The
second
representation
shows the approximate outline of the cardiac shadow. The anterior
portions of
the ra and rv have been removed.
Svc -superior vena cava ra-
right atrium.
Rv- right
ventricle. Vs-interventricular septum. pt- pulmonary trunk. Lpa- left
pulmonary
artery. Rpa- right pulmonary artery. a- aorta. aa- aortic arch. lv left
ventricle. lpv- left pulmonary vein
RH-
right hilum. LH- left hilum. AK - aortic knuckle. PA- pulmonary artery
Assessment of chamber size is
best done
by echocardiography
but the classic CXR signs of left atrial enlargement
are accurate (double right heart border, infilling of the
concavity between the aortic knuckle and the pulmonary artery and
splaying of
the carina).
The Hila
are
central mediastinal structures and
the hilum on
the side to which rotation has occurred may become obscured by the
heart
shadow. Note the anatomy of the pulmonary vessels is such that the left
hilum
is the higher. If this is reversed or even if they are at the same
level there
is likely to be some collapse somewhere.
7 Diaphragm
Normally at the level of the 10th. posterior rib but the position is very variable and height is dependant on a number of factors including radiographic technique. It is a thin structure (3mm) and if it appears to be thicker than this then the gas delineating its lower surface is probably in gut, rather than due to a pneumoperitoneum.
8 Lung
fields
Should be of similar density bilaterally. The basal arteries should look notably thicker and longer than those to the upper zones. The horizontal fissure should be at the level of the RT hilum and is normally very thin. Bronchial walls, if seen at all, should be thin and restricted to the perihilar regions. Any measurable thickness indicates peribronchial thickening.
The lateral is
sometimes helpful in the further
localisation of
pathology but has been largely superceded by CT.
It may identify small
effusions or basal pneumonia not seen on frontal CXRs. Posteriorly the
lung is darkest just above the diaphragm.
If not
then there is probably some basal pathology, either an effusion or
posterior
consolidation. The shadows of the humeri, glenoids and scapulae can
occasionally be misinterpreted. Look at the diaphragms. Note that you
can see
almost the whole length of the right one but not the anterior end of
the left
where it comes up against the heart.
Fig3 Normal lateral
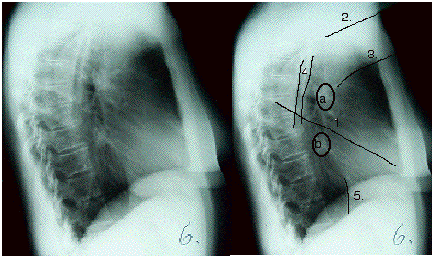
1. oblique fissure 2. humerus 3. soft tissue
of arm 4. scapulae
(anterior borders) 5. IVC (posterior border)
a Hila
b Confluence of pulmonary veins
-------------------
Abnormal
Lung shadows
Disease in the different
anatomical
divisions of the
lung give rise to specific appearances and distinction can usually be
made
between: Interstitial shadows, air space or alveolar shadowing (often
called
consolidation) and pleural abnormalities. All of these frequently
co-exist as
in; pneumonia, LVF and ARDS but one type of shadowing will usually
dominate.
Having decided which of these you are looking at it is then necessary
to match
up the clinical history with the type and distribution of the shadowing.
Interstitial
Lines and dots with variable
distribution. COAD
causes peribronchial
thickening, LVF tends to thicken up the peripheral interstitium (Kerley B lines) as well
as causing
peribronchial thickening. Malignant involvement (lymphangitis
cacinomatosa)
tends to cause central perihilar lines. Drug reactions are often
widespread and
may be associated with alveolar shadowing.
A
reticular pattern in the lower zones is typical of
fibrosing alveolitis. A pathognomic appearance of interstitial disease
is a
shaggy appearance to the heart or diaphragm due to adjacent
interstitial thickening.
Honeycombing is rare.
Fig4 Examples of Interstitial
disease.
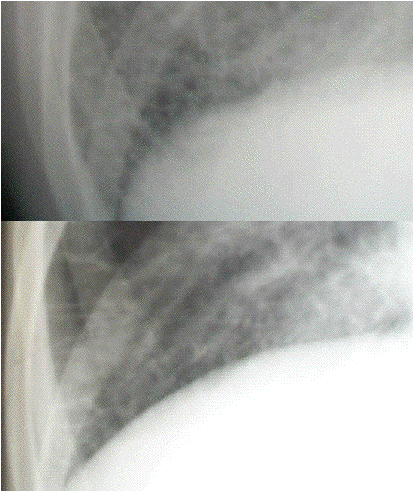
fig 4 Shows Close ups of two patients with interstitial shadowing the first has fibrosing alveolitis and shows the typical shaggy diaphragm. The second has interstitial pulmonary oedema and shows Kerley B lines. Note the relatively sharp diaphragm.
Alveolar
shadowing. 'Fluffy'
shadows tending towards
coalescence and fading away at the edges except where bounded by a
fissure
-shadowing with a sharp edge at a fissure or with a clear lobar or
segmental
distribution must be alveolar. It is due to fluid in the alveolar
spaces. Soft
tissue borders adjacent to the shadowing are lost but any air
containing spaces
within the shadowing will be outlined and air bronchograms are a
characteristic
feature. Distribution of the alveolar shadowing is helpful. A lobar
distribution is suggestive of pneumonia. Central positioning is
suggestive of
LVF or fluid overload. Widespread and changing alveolar shadowing may
indicate
opportunist, including fungal, infection.
Fig
5
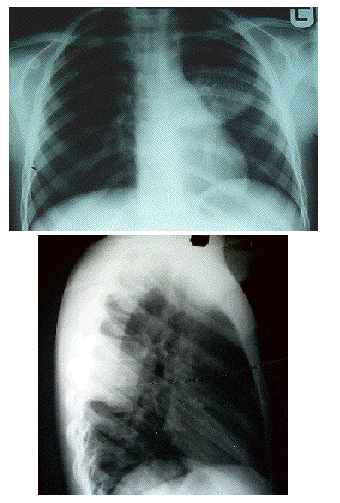
Fig 5 shows consolidation adjacent to the upper left heart border but
the heart
border remains visible. This means that the consolidation must be
posterior.
The lateral view shows the consolidation to be very posterior,
overlying the
vertebral bodies. The
consolidated
segment is below the level of the oblique fissure and therefore must be
in the
apical segment of the lower lobe. If it were above the oblique fissure
it would
be in the posterior segment of the upper lobe.
Pleural
fluid causes loss of the costophrenic angles
and a homogenous basal shadow with a concave upper border (on the erect
view);
there may be thickening of the horizontal fissure. Smaller amounts of
fluid may
be seen on the lateral in the posterior costophrenic recess.
Masses
Note the size and whether they are well or ill defined. Ill-defined, spiculated masses are usually primary tumours or possibly granulomas. Note any other lesions and any adjacent pathology (e.g. collapse or overlying rib destruction). Thick-walled cavitation suggests an abscess or tumour. Multiple cavities suggest abscesses or infarcts. Try to decide whether a lesion is pulmonary or pleural. En face pleural lesions tend to fade away at the edges. Lung and pleural masses up against the chest wall have fundamentally different shapes. Figure 6. Check the bones to exclude secondaries. Emphysema produces bullae, low flat diaphragms (below the anterior 7th rib), lack of peripheral vessels and an increase in the branch angles of the arteries. Other pathology (e.g. left ventricular failure) may be difficult to identify in the presence of emphysema.
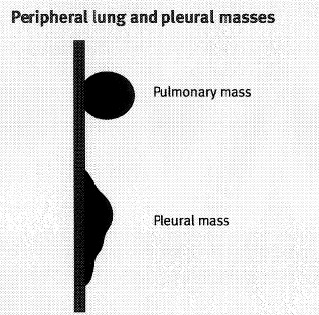 FIG6
FIG6


Asbestos related disease is predicted to increase considerably in the next few years. Asbestos exposure is indicated by thin liner pleural calcifications along the chest wall or diaphragm, asbestosis by associated changes similar to fibrosing alveolitis and mesothelioma by local pleural thickening and effusions. Figure 6 Isolated well defined pleural masses are more likely to represent pleural secondary deposits.
Emphysema
produces:
bullae, low flat diaphragms
(below the anterior 7th rib), lack
of peripheral
vessels and
an increase in the branch angles of the arteries. Other pathology e.g.
LVF may
be difficult to identify in the presence of emphysema.
--------------------------------
The ITU film
The problems associated with
supine and
portable
films are exacerbated by the condition of the patients in ITU. They are
usually
unable to cooperate and their lungs are often poorly compliant causing
unusual
appearances to familiar pathologies. The information gained from a
single ITU
film may therefore be limited counterbalanced by the availability of
serial,
usually daily, films allowing the progression of changes to be
recognised.
Abnormally
placed gas
Familiarity with the appearances of surgical emphysema and pneumomediastinum - recognised by air outlining the upper mediastinal structures is necessary as they may point to the presence of an unrecognised pneumothorax or possibly a pneumoperitoneum.
Fig 7
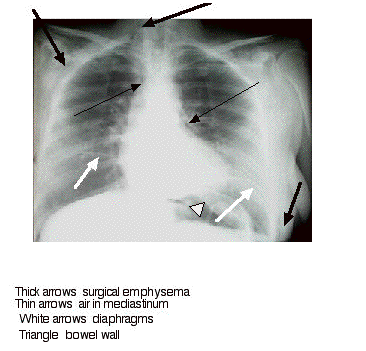
This shows a patient with a
massive
pneumoperitoneum
in whom the gas has tracked up behind the crura into the mediastinum
and out
through the thoracic inlet to cause surgical emphysema.
Pneumothorax in the supine ITU
patient
rarely
conforms to the classic appearances of a thin apical or upper zone line
with no
lung markings peripherally. A white line may be seen adjacent to the
mediastinum or roughly parallel to the chest wall or parts of the
mediastinum
may appear sharper than is usual.
There may be no radiological evidence of a pneumothorax and a high degree of
suspicion in
appropriate ITU patients is necessary.
Pneumopericardium is recognised
by the
presence of
air completely surrounding the heart shadow. It is usually benign
except for
the rare cases of tension but may point to the presence of serious
pathology
such as fistulae or infection.
Pleural fluid
in the supine or
even semi
erect patient lies
over the
posterior chest
wall and will not
have the familiar clear cut upper edge. Usually a veil of increased
density is
seen in the lower parts of the lung fields. It may be distinguished
from
alveolar shadowing because the basal pulmonary vessels may be seen and
by the
lack of an air bronchogram. However the two pathologies frequently
coexist.
Ultrasound shows effusions well and can be used to guide drains if
necessary. The
sudden appearance
of an effusion in a patient without fluid overload may suggest a
haemothorax.
Loculated effusions may show as well defined masses or thickening of
fissures
and should raise suspicions of an empyema or bleeding. A fluid level
going
straight across the lung field on an erect film indicates fluid plus
air in the
pleural space.
Alveolar shadows
can be associated
with:
Infection, Pulmonary oedema, aspiration, contusion, and ARDS.
Infections. A lobar or segmental distribution suggests lobar pneumonia. More diffuse shadowing in an immunocompromised patient will suggest opportunist infection including fungi. Persistent upper zone shadowing in association with cavitation and fibrosis may mean TB.
Cavitation anywhere could mean
specific
organisms
such as: TB, staph. or pneumococcus and multiple 1-2cm cavities raise
the
possibility of septic emboli.
Lung Oedema - diffuse if
associated with:
LVF, fluid
overload and fresh water drowning. May be localised following
aspiration and
associated with other changes including atelectasis.
Contusion may be localised to
the site of
injury and
show some progression for some days following the injury. Pulmonary
haemorrhage
can be diffuse and indistinguishable from pulmonary oedema.
ARDS can mimic almost any other
acute
condition.
Radiologically it is best identified by its relentless progression from
central
pulmonary alveolar shadowing to more generalised and persistent
coverage of the
whole lung.
Atelectasis can vary from
complete
collapse of a
lung as evidenced by massive mediastinal shift to the side of the
collapse to
small often temporary white lines in subsegmental atelectasis. It is
only
rarely associated with demonstrable mucous plugging. The various
appearances of
lobar collapse will be dealt with elsewhere.
Lines
etc
Radiographers should be made
aware of the
indication
for a chest X-ray done for a specific line or tube as they may be able
to vary
the technique accordingly and may repeat the film automatically if the
line is
not shown well. Anaesthetists may like to know that ultrasound
measurements have
shown
that raising the patient's feet produces just as much jugular
vein dilatation
as tipping the patient head down.
An endotracheal tube should be
at least
5cm above
the carina as flexion/extension of the head can move it by as much as 4
cms.
A small pneumomediastinum or
surgical
emphysema is
not uncommon following the insertion of a tracheostomy tube.
On the Chest X ray a subclavian
line can
be expected
to be entering the subclavian vein at the lateral edge of the first
rib, the
brachiocephalic vein at the sternoclavicular joint and the SVC at the
first
anterior intercostal space. Commonest malplacements are upwards into
the
jugular vein and across the midline into the opposite brachiocephalic
vein. A
medially placed deviation at its lower end may indicate placement into
t Beware of the line which shows
a small
kink or even
a gentle deviation at its distal end. It may be up against the vessel
wall with
the potential of erosion. This particularly applies to a large catheter
in the relatively thin pulmonary arteries, particularly in the presence
of pulmonary
hypertension. The rare complication of catheter breakage should be
dealt with
as soon as possible, preferably by an interventional radiologist. Such
large
emboli can drift more peripherally with time and can erode vessels
rapidly,
particularly in the pulmonary circulation. Pacemakers. Most single-wire
pacemaker wires are
inserted into
the subclavian vein and directed into the apex of the right ventricle.
A
lateral view will show the distal end of the correctly placed wire
coursing
anteriorly. The course of the wires should be smooth on both the AP and
lateral
views and any localised kinks be viewed with suspicion. The commonest
site of
wire fracture is between the clavicle and the first rib, usually well
seen on
the frontal view. Nasogastric tubes have holes
along their
distal 10
cm and need to be well into the stomach to stop gastro oesophageal
reflux
occurring via these holes. They can enter the bronchial tree but
bizarre
looking 'malplacements' are usually due to the presence
of a hiatus hernia
rather than to oesophageal rupture. FIG 8 --> FIG 8 Further reading Seminars in Radiology 1997
volXXX11 Diagnostic Radiology, Grainger
and
Allison,
Churchill Livingston Vol1 Sections 2&3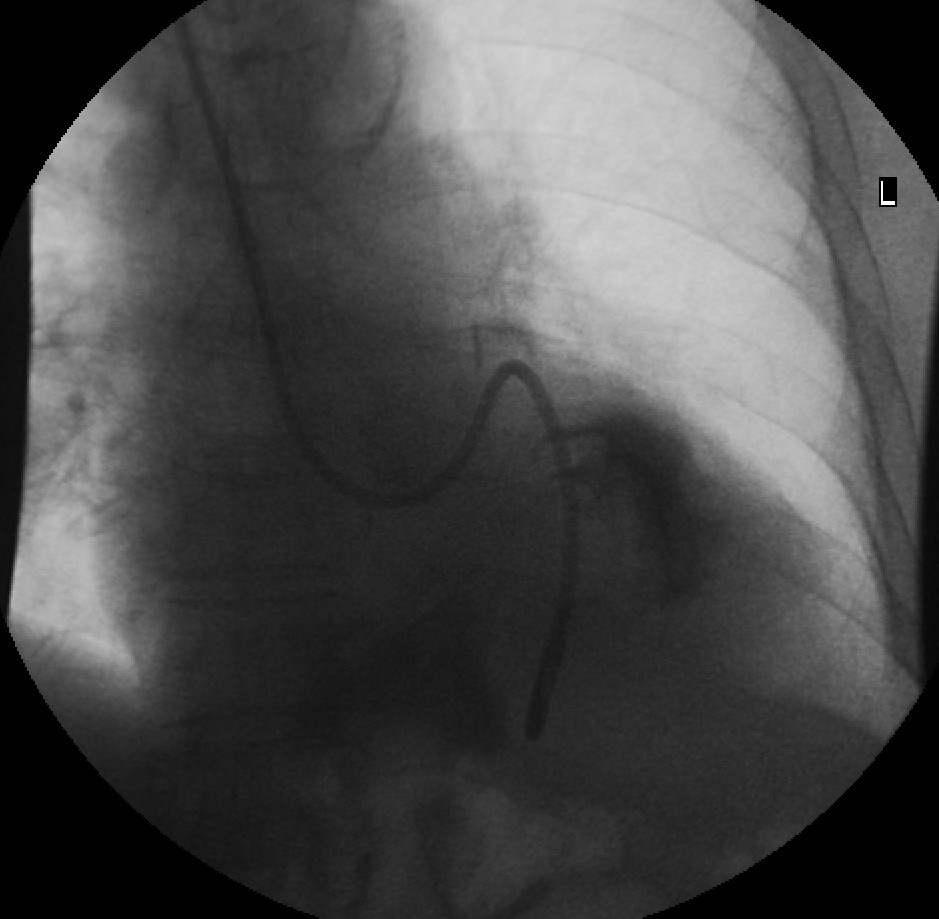
This patient had her NG tube replaced 3 times on the ward before being
sent down to X-Ray to have the tube insertion under fluroscopy because
of ‘repeated aspiration’ of the tube. Note that the
left
main bronchus is demonstrated well above the tube as it deviates to the
left. A little contrast has been injected into the tube to show the
incarcerated hiatus hernia.